Compounded
Semaglutide
Compounded semaglutide is different from branded products such as Ozempic® and Wegovy®. Eligibility is determined by a licensed provider based on your medical history. Individual results vary.

What is Semaglutide?
Semaglutide is a GLP-1 (glucagon-like peptide-1) receptor agonist — a class of medication that mimics a hormone your gut naturally produces after eating. It was originally developed by Novo Nordisk and brought to market as Ozempic® (for type 2 diabetes) and Wegovy® (for chronic weight management).
At AvataCore, licensed providers may prescribe compounded semaglutide prepared at state-licensed U.S. compounding pharmacies. Compounded medications are different from branded products. Formulation, strength, inactive ingredients, and clinical effects may differ from branded versions. See our Medication Options disclosure for full details.
Drug class
GLP-1 receptor agonist
Original brand
Ozempic® / Wegovy® (Novo Nordisk)
Administration
Once-weekly subcutaneous injection
How Semaglutide Works
Appetite Regulation
Semaglutide activates GLP-1 receptors in the hypothalamus — the brain's hunger control center — sending satiety signals that meaningfully reduce appetite and food intake.
Slows Gastric Emptying
By slowing the rate at which the stomach empties, semaglutide prolongs the feeling of fullness after meals, reducing the urge to snack or overeat.
Reduces Cravings
Beyond physical hunger, semaglutide also acts on reward pathways linked to food cravings, helping patients feel less compelled by high-calorie, high-fat foods.
Dosing Schedule
A gradual dose escalation over 16–20 weeks allows your body to adjust, minimizing side effects while building to full therapeutic effect.
Initiation dose — allows body to adapt
First escalation — appetite reduction increases
Therapeutic range for most patients
Optional escalation if tolerated
Maximum dose — per provider discretion
* Dosing is individualized. Your prescribing provider may adjust based on response and tolerability.
Common Side Effects
Most side effects are gastrointestinal, mild-to-moderate, and temporary — most improving within the first few weeks at each dose level.
Most Common (≥10% of patients)
Nausea
Tip: Eat smaller portions; avoid fatty or spicy foods; take in the evening
Constipation
Tip: Increase fiber and fluid intake; gentle walking helps
Diarrhea
Tip: Usually transient; stay hydrated
Fatigue
Tip: Common early on; improves as body adjusts
Less Common — Seek Care If Severe
Vomiting
If persistent, contact your care team
Abdominal pain
Severe or persistent pain warrants immediate evaluation
Headache
Typically resolves; ensure adequate hydration
Heart rate changes
Inform your provider of any palpitations
BOXED WARNING — Risk of Thyroid C-Cell Tumors
Compounded semaglutide/tirzepatide carries the same boxed warning as FDA-approved formulations. GLP-1 receptor agonists caused thyroid C-cell tumors in rodents. It is unknown whether these medications cause thyroid C-cell tumors, including medullary thyroid carcinoma (MTC), in humans. These medications are contraindicated in patients with a personal or family history of MTC or in patients with Multiple Endocrine Neoplasia syndrome type 2 (MEN 2).
Clinical Results
Semaglutide's efficacy for weight management is supported by the landmark STEP (Semaglutide Treatment Effect in People with obesity) clinical trial program.
14.9%
Mean body weight reduction
STEP 1 trial at 68 weeks
2.4 mg
Weekly dose studied
vs. 2.4% with placebo
1,961
Participants in STEP 1
Adults with obesity or overweight + comorbidity
STEP 1 Trial Summary
Published in the New England Journal of Medicine (2021), the STEP 1 trial was a 68-week, randomized, double-blind, placebo-controlled trial. Adults with a BMI ≥30 (or ≥27 with weight-related comorbidities) received once-weekly semaglutide 2.4 mg or placebo alongside lifestyle intervention. The semaglutide group achieved a mean weight loss of 14.9% vs. 2.4% for placebo — a statistically significant 12.4 percentage-point difference. Over 86% of participants receiving semaglutide lost ≥5% of body weight.
Reference: Wilding JPH et al. Once-Weekly Semaglutide in Adults with Overweight or Obesity. N Engl J Med. 2021;384(11):989-1002.
Injection Site Guide
Semaglutide is injected subcutaneously (just under the skin) once weekly. Rotate sites with each injection to avoid tissue buildup.
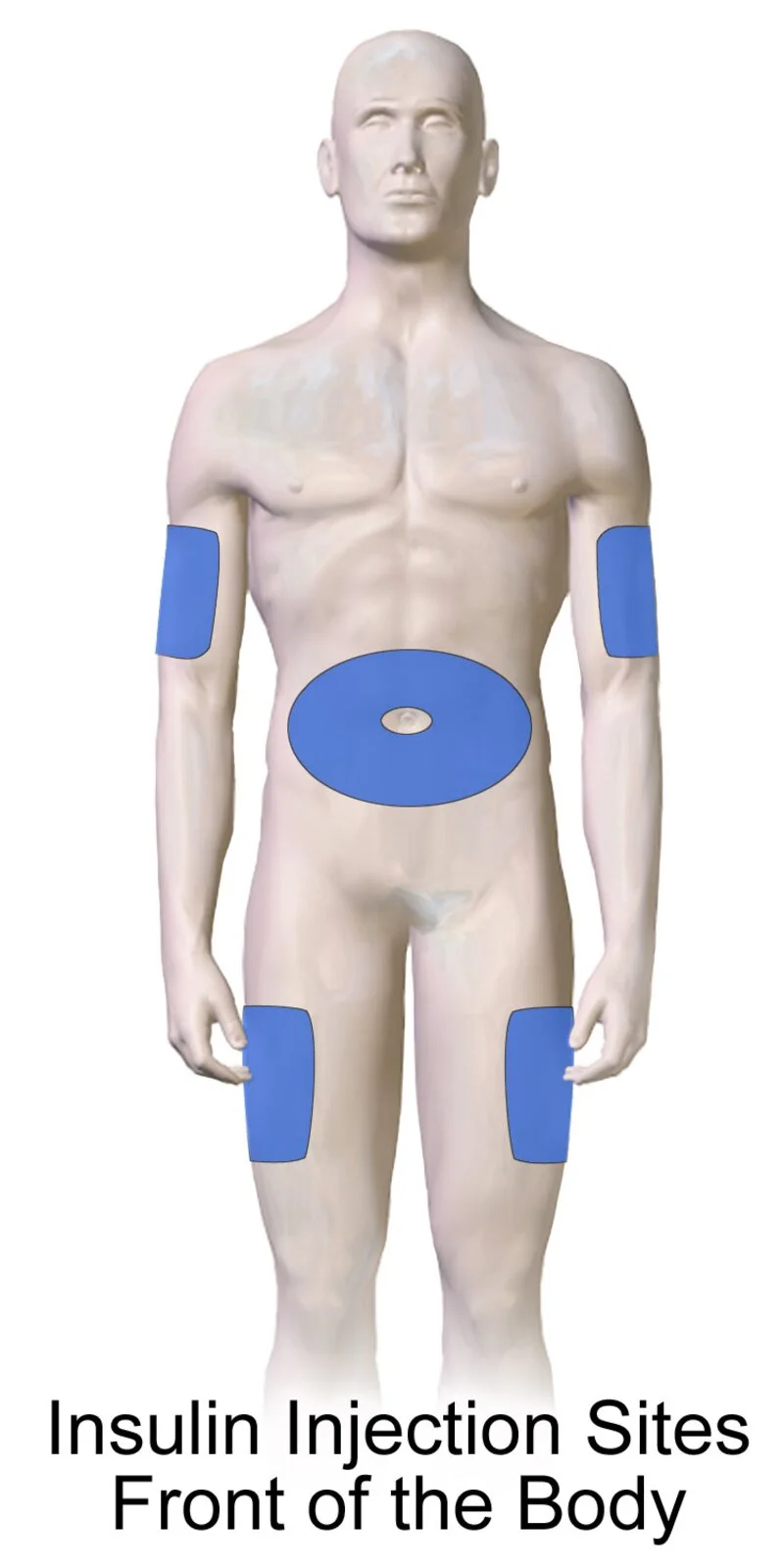
Abdomen
Preferred site. Inject at least 2 inches from navel. Easy self-administration.
Outer Thigh
Front of thigh, midway between knee and hip. Good alternative to abdomen.
Upper Arm
Outer aspect only. May require assistance. Slightly less predictable absorption.
Rotate injection sites weekly. Do not inject into skin that is tender, bruised, scarred, or hardened. Your care team will provide step-by-step video instructions.
Starting at $149
Everything included — consultation, medication, supplies, and ongoing provider support.
No hidden fees. Cancel anytime.
*Results not guaranteed. Provider consultation required.
Semaglutide Questions Answered
What is compounded semaglutide?
How is compounded semaglutide different from Ozempic® or Wegovy®?
How quickly will I see results?
What if I experience nausea?
Can I stop taking semaglutide once I reach my goal weight?
How is semaglutide used in patients who also have diabetes?
Brand-Name Reference
You may know these brand-name options
AvataCore does not dispense these. Pricing is publicly available retail. Learn why we use compounded medications instead.
Starting at $1,579
Wegovy® Tablets
Semaglutide · Daily tablet · Novo Nordisk
Wegovy® is a registered trademark of Novo Nordisk. Not sold by AvataCore.
Starting at $1,579
Wegovy® Injection
Semaglutide · Weekly injection · Novo Nordisk
Wegovy® is a registered trademark of Novo Nordisk. Not sold by AvataCore.
Starting at $1,249
Mounjaro® Injection
Tirzepatide · Weekly injection · Eli Lilly
Mounjaro® is a registered trademark of Eli Lilly. Not sold by AvataCore.

Starting at $1,249
Zepbound® Injection
Tirzepatide · Weekly injection · Eli Lilly
Zepbound® is a registered trademark of Eli Lilly. Not sold by AvataCore.

Starting at $1,149
Ozempic® Injection
Semaglutide · Weekly injection · Novo Nordisk
Ozempic® is a registered trademark of Novo Nordisk. Not sold by AvataCore.
Brand-name medications shown for reference. AvataCore does not directly dispense Ozempic®, Wegovy®, Mounjaro®, or Zepbound®. Pricing reflects publicly available retail estimates and is subject to change. Compounded medications are different from FDA-approved branded products. AvataCore is not affiliated with Novo Nordisk or Eli Lilly.
Ready to get started?
Complete a free eligibility assessment. A licensed provider will review your profile and recommend a personalized treatment plan — no office visit required.
Take the Quiz →