Compounded
Tirzepatide
The dual GIP/GLP-1 agonist in Mounjaro® and Zepbound®. In the SURMOUNT-1 trial, adults taking the 15 mg dose lost an average of 20.9% of their body weight at 72 weeks.*

What is Tirzepatide?
Tirzepatide is a first-in-class dual GIP/GLP-1 receptor agonist — a medication that simultaneously activates two different gut hormone receptors involved in appetite, metabolism, and insulin regulation. It was developed by Eli Lilly and brought to market as Mounjaro® (for type 2 diabetes) and Zepbound® (for chronic weight management).
By targeting both the GLP-1 and GIP pathways, tirzepatide has demonstrated greater average weight loss than GLP-1–only medications in head-to-head clinical trials. In the SURMOUNT-1 trial, adults taking tirzepatide 15 mg lost an average of 20.9% of their body weight over 72 weeks.*
Drug class
Dual GIP/GLP-1 receptor agonist
Original brand
Mounjaro® / Zepbound® (Eli Lilly)
Administration
Once-weekly subcutaneous injection
How Tirzepatide Works
Tirzepatide's dual-receptor mechanism is what sets it apart — activating both GLP-1 and GIP pathways produces synergistic effects greater than either pathway alone.
- Suppresses appetite via hypothalamus
- Slows gastric emptying (prolongs satiety)
- Reduces food cravings and reward-driven eating
- Improves insulin secretion after meals
- Enhances fat metabolism and energy expenditure
- Potentiates GLP-1 effects on appetite
- Improves glucose metabolism
- May reduce GLP-1 side effects (nausea)
The result: more powerful hunger suppression, greater metabolic benefits, and superior weight loss outcomes compared to GLP-1–only therapy.
Dosing Schedule
Tirzepatide starts at a low dose and escalates gradually over 20 weeks to the maintenance dose, minimizing side effects while building to full therapeutic effect.
Initiation dose — lowest effective dose for tolerance
First escalation — significant appetite reduction begins
Therapeutic range for many patients
Continued escalation if tolerated
High therapeutic dose
Maximum dose — per provider discretion
* Dosing is individualized. Your prescribing provider may adjust based on your response, tolerability, and weight-loss goals.
Common Side Effects
Tirzepatide's side effect profile is similar to semaglutide — primarily gastrointestinal and typically mild-to-moderate during dose escalation.
Most Common (≥10% of patients)
Nausea
Tip: Most common during escalation; eat smaller portions and avoid fatty foods
Diarrhea
Tip: Usually transient; stay well-hydrated
Constipation
Tip: Increase fiber intake; adequate hydration helps
Vomiting
Tip: Contact care team if persistent or severe
Less Common — Seek Care If Severe
Abdominal pain
Severe or persistent pain warrants immediate evaluation
Decreased appetite / fatigue
Monitor nutrition; ensure adequate caloric intake
Injection site reactions
Redness or swelling; rotate injection sites each week
Heart rate changes
Inform your provider of any palpitations
BOXED WARNING — Risk of Thyroid C-Cell Tumors
Compounded semaglutide/tirzepatide carries the same boxed warning as FDA-approved formulations. GLP-1 receptor agonists caused thyroid C-cell tumors in rodents. It is unknown whether these medications cause thyroid C-cell tumors, including medullary thyroid carcinoma (MTC), in humans. These medications are contraindicated in patients with a personal or family history of MTC or in patients with Multiple Endocrine Neoplasia syndrome type 2 (MEN 2).
Clinical Results
Tirzepatide's efficacy is supported by the SURMOUNT clinical trial program, published in the New England Journal of Medicine.
20.9%
Mean body weight reduction
SURMOUNT-1 trial, 15mg dose, treatment-policy estimand (ITT) at 72 weeks
91%
Lost ≥5% body weight
vs. 35% with placebo
2,539
Participants in SURMOUNT-1
Adults with obesity or overweight + comorbidity
Tirzepatide vs. Semaglutide — Weight Loss Comparison
SURMOUNT-1 Trial Summary
Published in the New England Journal of Medicine (2022), the SURMOUNT-1 trial was a 72-week, randomized, double-blind, placebo-controlled trial of 2,539 adults with obesity (BMI ≥30) or overweight with weight-related comorbidities. Participants received tirzepatide 5 mg, 10 mg, or 15 mg weekly. The highest-dose group (15 mg) achieved a mean weight reduction of 20.9%. 91% of tirzepatide participants lost ≥5% body weight vs. 35% in the placebo group.
Reference: Jastreboff AM et al. Tirzepatide Once Weekly for the Treatment of Obesity. N Engl J Med. 2022;387(3):205-216.
Injection Site Guide
Tirzepatide is administered as a once-weekly subcutaneous injection. Rotating sites prevents tissue irritation and ensures consistent absorption.
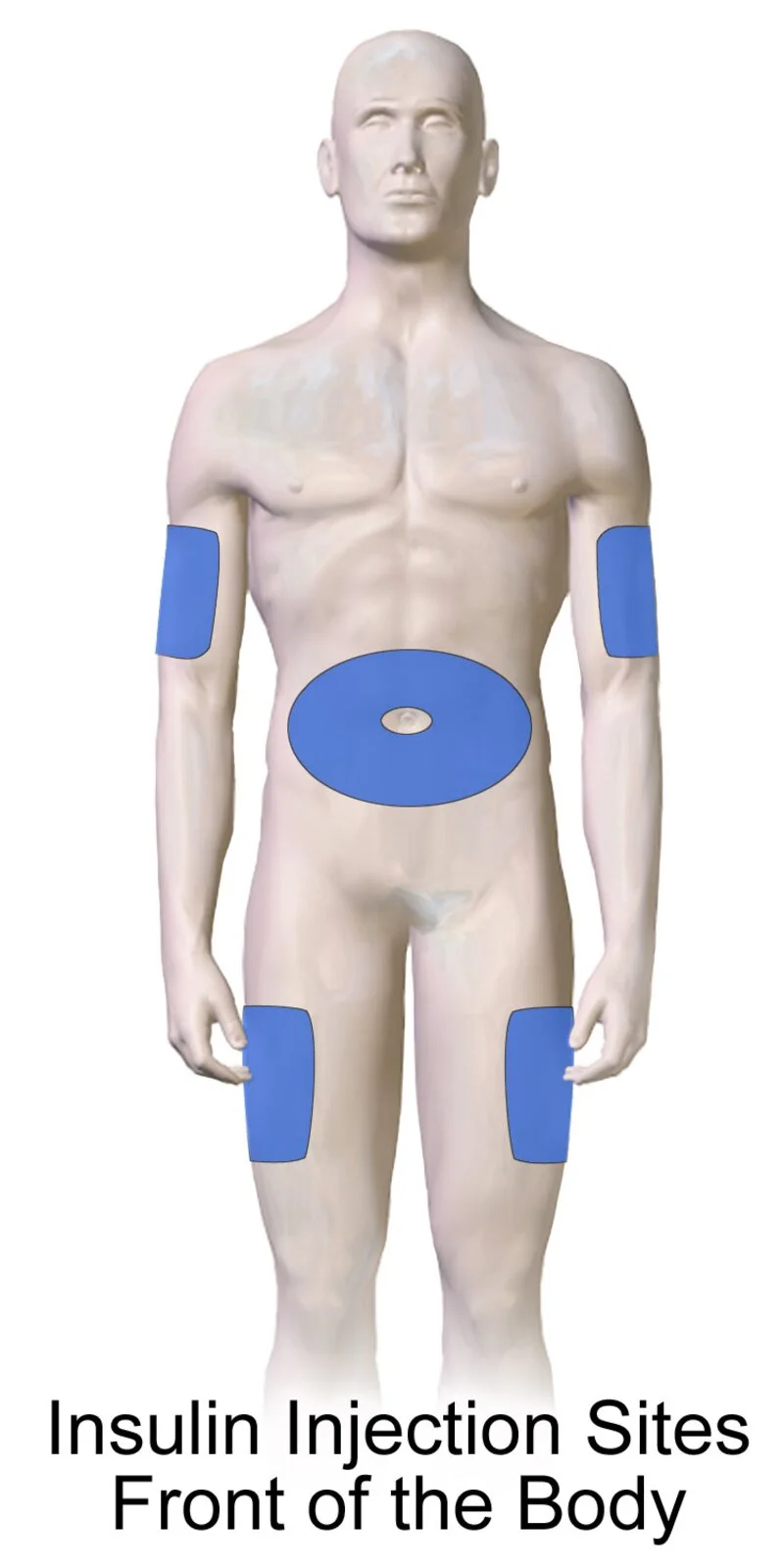
Abdomen
Preferred site. Inject at least 2 inches from navel. Easiest for self-administration.
Outer Thigh
Front of thigh, midway between knee and hip. Good alternative to abdomen.
Upper Arm
Outer aspect only. May require assistance. Slightly less predictable absorption.
Rotate injection sites weekly. Do not inject into skin that is tender, bruised, scarred, or hardened. Step-by-step video instructions are provided to all patients.
Starting at $199.
Everything included — consultation, dual-action medication, supplies, and ongoing provider support.
No hidden fees. Cancel anytime.
*Results not guaranteed. Provider consultation required.
Tirzepatide Questions Answered
What is compounded tirzepatide?
How is tirzepatide different from semaglutide?
How quickly will I see results with tirzepatide?
What side effects should I expect?
How is tirzepatide used in patients who also have type 2 diabetes?
Who is tirzepatide most suitable for?
Brand-Name Reference
You may know these brand-name options
AvataCore does not dispense these. Pricing is publicly available retail. Learn why we use compounded medications instead.
Starting at $1,579
Wegovy® Tablets
Semaglutide · Daily tablet · Novo Nordisk
Wegovy® is a registered trademark of Novo Nordisk. Not sold by AvataCore.
Starting at $1,579
Wegovy® Injection
Semaglutide · Weekly injection · Novo Nordisk
Wegovy® is a registered trademark of Novo Nordisk. Not sold by AvataCore.
Starting at $1,249
Mounjaro® Injection
Tirzepatide · Weekly injection · Eli Lilly
Mounjaro® is a registered trademark of Eli Lilly. Not sold by AvataCore.

Starting at $1,249
Zepbound® Injection
Tirzepatide · Weekly injection · Eli Lilly
Zepbound® is a registered trademark of Eli Lilly. Not sold by AvataCore.

Starting at $1,149
Ozempic® Injection
Semaglutide · Weekly injection · Novo Nordisk
Ozempic® is a registered trademark of Novo Nordisk. Not sold by AvataCore.
Brand-name medications shown for reference. AvataCore does not directly dispense Ozempic®, Wegovy®, Mounjaro®, or Zepbound®. Pricing reflects publicly available retail estimates and is subject to change. Compounded medications are different from FDA-approved branded products. AvataCore is not affiliated with Novo Nordisk or Eli Lilly.
Not sure which medication is right for you?
Our licensed providers will review your health profile and recommend semaglutide or tirzepatide based on your specific goals and medical history.